What Is Retatrutide?
Retatrutide (also known as LY3437943) is an investigational peptide drug developed by Eli Lilly and Company that represents a new frontier in metabolic disease treatment. Unlike existing GLP-1 receptor agonists such as semaglutide (Ozempic/Wegovy) or dual agonists like tirzepatide (Mounjaro/Zepbound), retatrutide is the world's first triple hormone receptor agonist — simultaneously activating three key metabolic receptors: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide 1 (GLP-1), and glucagon receptors.
This triple mechanism of action earned retatrutide the informal designation as a "GLP-3 agonist" in some media coverage, though this is technically a misnomer — it activates three different receptor types rather than a single "GLP-3" receptor.
The Development Story
Retatrutide emerged from Eli Lilly's incretin research program, building on the success of tirzepatide (a dual GIP/GLP-1 agonist). The key innovation was adding glucagon receptor (GCGR) agonism to the existing dual-agonist framework. Preclinical studies published in Cell Metabolism in 2022 demonstrated that the triple-agonist approach produced superior weight loss and metabolic improvements compared to dual agonists in animal models [1].
The first-in-human Phase 1b clinical trial was published in The Lancet in 2022, establishing that retatrutide had an acceptable safety profile and pharmacokinetics suitable for once-weekly dosing in people with type 2 diabetes [2].
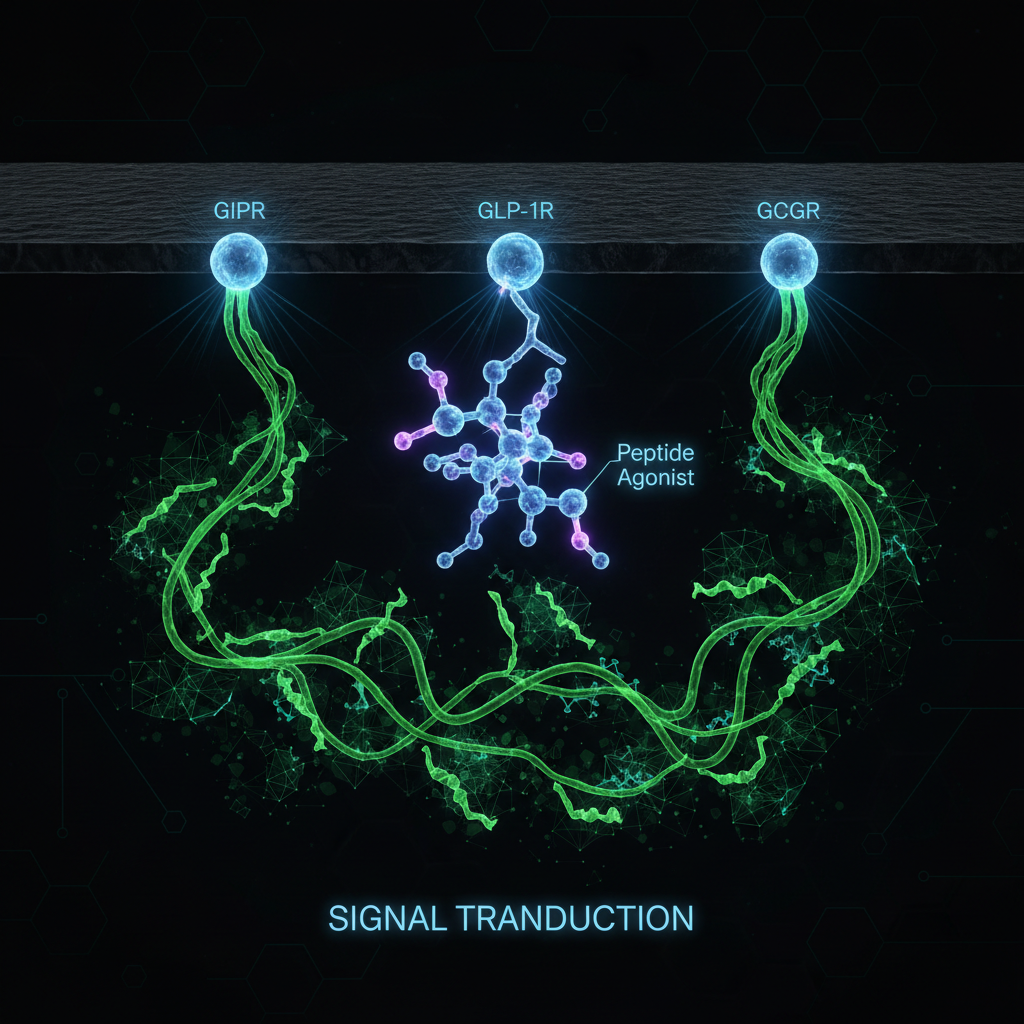
How the Three Receptors Work Together
GLP-1 Receptor Activation
GLP-1 receptor agonism is the foundation of retatrutide's action. It reduces appetite by acting on brain centers that control hunger and satiety, slows gastric emptying to promote fullness, and enhances glucose-dependent insulin secretion from pancreatic beta cells. This mechanism is shared with semaglutide and liraglutide.
GIP Receptor Activation
Glucose-dependent insulinotropic polypeptide (GIP) receptor activation complements GLP-1 effects. GIP enhances insulin secretion, may improve fat metabolism, and appears to have central nervous system effects that reduce food intake. The combination of GIP and GLP-1 agonism — as seen in tirzepatide — produces greater weight loss than GLP-1 alone.
Glucagon Receptor Activation
The glucagon receptor component is what makes retatrutide unique. Glucagon traditionally raises blood sugar by stimulating hepatic glucose production, which might seem counterproductive in a diabetes drug. However, glucagon receptor activation also increases energy expenditure, promotes lipid oxidation (fat burning), and reduces liver fat content. In the context of retatrutide's balanced triple-agonist profile, the metabolic benefits of glucagon receptor activation — particularly increased energy expenditure and liver fat reduction — outweigh the glucose-raising effects, which are offset by the concurrent GLP-1 and GIP activity [3].
Structural Biology
In 2024, researchers used cryo-electron microscopy (cryo-EM) to determine the three-dimensional structures of retatrutide bound to each of its three target receptors — GLP-1R, GIPR, and GCGR. This structural analysis revealed how a single peptide molecule can activate three distinct receptors, providing insights into the molecular basis of retatrutide's triple agonism and informing future drug design efforts [4].
Current Development Status
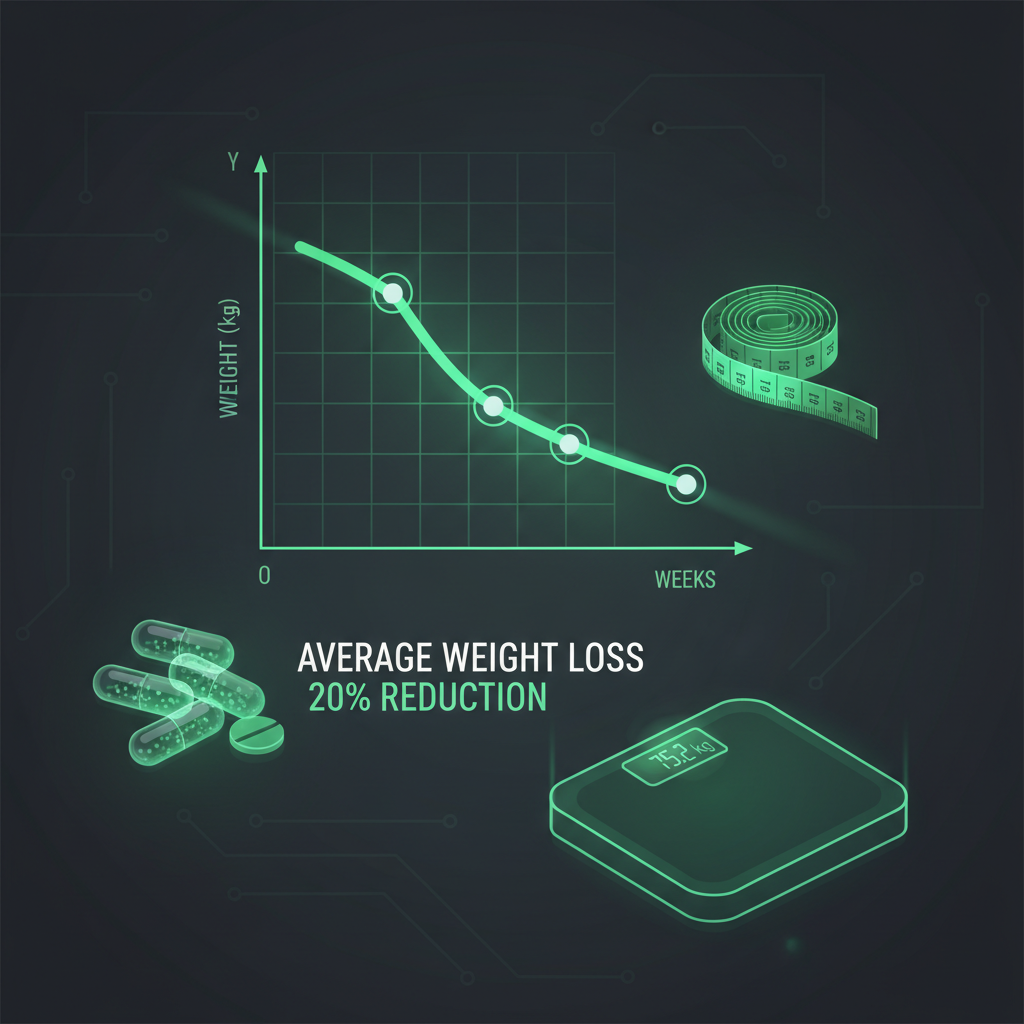
Retatrutide is currently in Phase 3 clinical development across multiple large-scale trials collectively known as the TRANSCEND program. These include trials in obesity (TRANSCEND-OB), type 2 diabetes (TRANSCEND-T2D), cardiovascular outcomes (TRANSCEND-CV), and metabolic liver disease. Early Phase 3 results from TRANSCEND-T2D-1 showed A1C reductions of 1.7% to 2.0% and weight loss up to 16.8% at 40 weeks.
References
-
Coskun T, et al. "LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss: from discovery to clinical proof of concept." Cell Metabolism. 2022;34(9):1234-1247. PubMed: 35985340
-
Urva S, et al. "LY3437943, a novel triple GIP, GLP-1, and glucagon receptor agonist in people with type 2 diabetes: a phase 1b, multicentre, double-blind, placebo-controlled, randomised, multiple-ascending-dose trial." The Lancet. 2022;400(10366):1869-1881. PubMed: 36354040
-
Doggrell SA. "Is retatrutide (LY3437943), a GLP-1, GIP, and glucagon receptor agonist a step forward in the treatment of diabetes and obesity?" Expert Opinion on Investigational Drugs. 2023;32(5):377-381. PubMed: 37086147
-
Li W, et al. "Structural insights into the triple agonism at GLP-1R, GIPR and GCGR manifested by retatrutide." Cell Discovery. 2024;10:73. DOI: 10.1038/s41421-024-00700-0