The Science Behind Triple Agonism
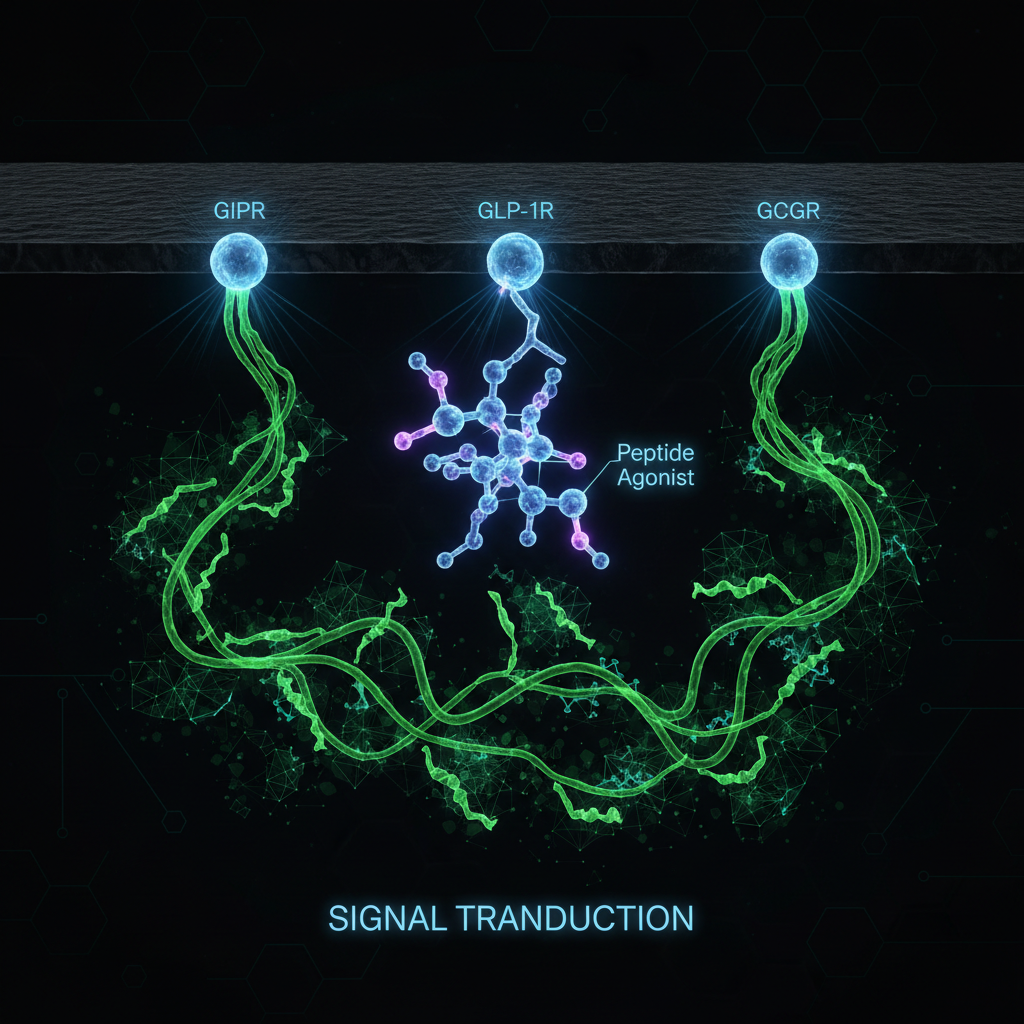
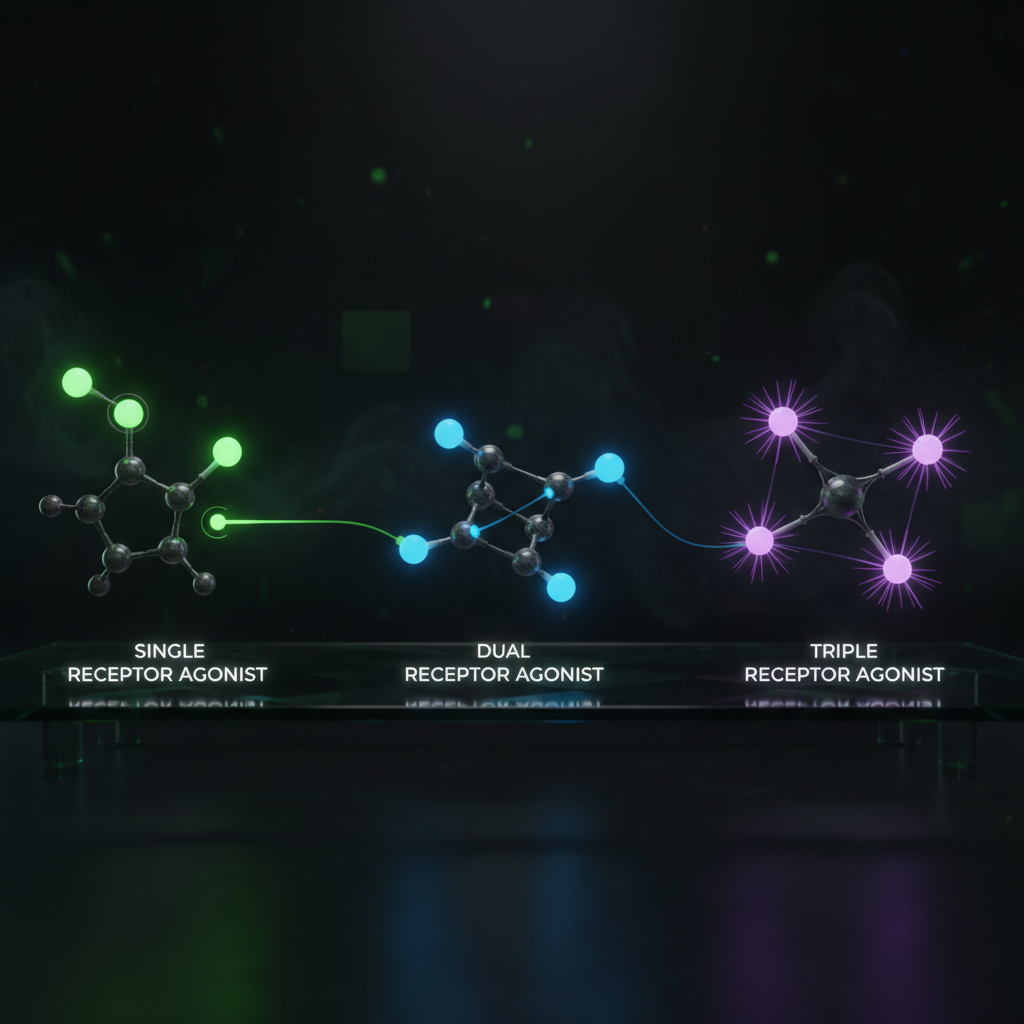
Retatrutide represents a paradigm shift in metabolic pharmacology. Rather than targeting a single pathway, it engages three complementary receptor systems simultaneously. Understanding how each receptor contributes to the overall therapeutic effect reveals why this approach produces superior outcomes compared to single or dual agonists.
GLP-1 Receptor: The Appetite Suppressor
Glucagon-like peptide 1 (GLP-1) is an incretin hormone naturally released from intestinal L-cells after eating. The GLP-1 receptor is expressed in the pancreas, brain, gastrointestinal tract, heart, and kidneys.
Key effects of GLP-1R activation:
- Central appetite suppression: GLP-1R in the hypothalamus and brainstem reduces hunger signals and promotes satiety
- Delayed gastric emptying: Slows the rate at which food leaves the stomach, prolonging the feeling of fullness
- Glucose-dependent insulin secretion: Stimulates insulin release only when blood glucose is elevated, reducing hypoglycemia risk
- Glucagon suppression: Reduces inappropriate glucagon secretion in the fasting state
- Beta-cell preservation: May protect pancreatic beta cells from apoptosis
GLP-1 receptor agonism is the most well-validated component, with decades of clinical evidence from drugs like liraglutide, semaglutide, and exenatide [1].
GIP Receptor: The Metabolic Amplifier
Glucose-dependent insulinotropic polypeptide (GIP) is another incretin hormone, released from intestinal K-cells. For decades, GIP was considered a less promising drug target because its insulinotropic effect is blunted in type 2 diabetes. However, the success of tirzepatide revealed that GIP receptor activation provides substantial benefits when combined with GLP-1 agonism.
Key effects of GIPR activation:
- Enhanced insulin secretion: Amplifies glucose-dependent insulin release in concert with GLP-1
- Central nervous system effects: GIPR in the brain contributes to appetite regulation and may enhance the anorectic effects of GLP-1
- Adipose tissue effects: GIP influences fat metabolism and may promote healthier fat distribution
- Bone metabolism: GIP has been shown to reduce bone resorption, potentially mitigating weight-loss-associated bone density loss
The combination of GIP and GLP-1 agonism produces greater weight loss and glycemic improvement than either alone, as demonstrated by tirzepatide's clinical success [2].
Glucagon Receptor: The Energy Expenditure Booster
The glucagon receptor (GCGR) is the most novel and controversial component of retatrutide's triple-agonist profile. Glucagon is traditionally viewed as a counter-regulatory hormone that raises blood sugar — seemingly counterproductive in a diabetes drug. However, glucagon receptor activation provides several metabolic benefits that are particularly valuable in the context of obesity treatment.
Key effects of GCGR activation:
- Increased energy expenditure: Glucagon stimulates thermogenesis and increases resting metabolic rate, promoting caloric burn even at rest
- Enhanced lipid oxidation: Promotes the breakdown and burning of stored fat, particularly in the liver
- Hepatic fat reduction: Directly reduces liver fat content by stimulating fatty acid oxidation in hepatocytes — this is retatrutide's most distinctive advantage
- Amino acid metabolism: Glucagon influences amino acid catabolism, which may have implications for body composition
The glucose-raising effect of glucagon is effectively counterbalanced by the simultaneous GLP-1 and GIP receptor activation, which enhance insulin secretion and suppress inappropriate glucagon release. This creates a metabolic environment where the beneficial effects of glucagon (energy expenditure, fat burning) are preserved while the hyperglycemic risk is mitigated [3].
Structural Biology: How One Molecule Activates Three Receptors
A landmark 2024 study used cryo-electron microscopy (cryo-EM) to solve the three-dimensional structures of retatrutide bound to each of its three target receptors. This structural analysis revealed that retatrutide adopts slightly different conformations when binding to GLP-1R, GIPR, and GCGR, allowing a single peptide molecule to effectively engage all three receptor types.
The cryo-EM structures showed that retatrutide's peptide backbone makes critical contacts with conserved residues in the extracellular domain and transmembrane core of each receptor, while specific side-chain interactions differ between receptors, explaining the molecule's balanced triple-agonist activity [4].
Synergistic Effects
The three receptor systems do not simply add their effects — they interact synergistically:
- GLP-1 + GIP → Enhanced appetite suppression and insulin secretion beyond either alone
- GLP-1 + Glucagon → GLP-1 offsets glucagon's hyperglycemic effect while glucagon adds energy expenditure
- GIP + Glucagon → GIP's metabolic effects complement glucagon's thermogenic and lipolytic actions
- All three together → The complete triple-agonist profile produces the most comprehensive metabolic improvement: reduced intake, increased expenditure, improved glycemia, and dramatic liver fat reduction
Gastric Emptying Effects
A dedicated pharmacological study examined retatrutide's effects on gastric emptying, finding dose-dependent delays consistent with GLP-1 receptor activation. This delayed gastric emptying contributes to the prolonged satiety and reduced food intake observed in clinical trials [5].
References
-
Doggrell SA. "Is retatrutide (LY3437943), a GLP-1, GIP, and glucagon receptor agonist a step forward in the treatment of diabetes and obesity?" Expert Opinion on Investigational Drugs. 2023;32(5):377-381. PubMed: 37086147
-
Rosenstock J, et al. "Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes." The Lancet. 2023;402(10401):529-544. PubMed: 37385280
-
Coskun T, et al. "LY3437943, a novel triple glucagon, GIP, and GLP-1 receptor agonist for glycemic control and weight loss." Cell Metabolism. 2022;34(9):1234-1247. PubMed: 35985340
-
Li W, et al. "Structural insights into the triple agonism at GLP-1R, GIPR and GCGR manifested by retatrutide." Cell Discovery. 2024;10:73. DOI: 10.1038/s41421-024-00700-0
-
"The novel GIP, GLP-1 and glucagon receptor agonist retatrutide delays gastric emptying." Diabetes, Obesity and Metabolism. 2023;25(11):3308-3316. PubMed: Referenced in DOM